ECG-based Potassium Measurement is Unaffected by Errors in Blood Potassium Measurement during Hemodialysis
Zachi Attia, Paul A Friedman, Dorothy J Ladewig, Guarav C Satam, Jennifer L Dugan, Yehu Sepir, Dan Sadot, Amir Geva, Virend K. Somers, Samuel J. Asirvatham, Michael J Ackerman, Kevin E Bennet, Bhupinder Singh and John J Dillon
DOI10.21767/2472-5056.100064
Zachi Attia1, Paul A Friedman1, Dorothy J Ladewig2, Guarav C Satam2, Jennifer L Dugan1, Yehu Sepir3, Dan Sadot3, Amir Geva3, Virend K. Somers1, Samuel J. Asirvatham1, Michael J Ackerman1, Kevin E Bennet4, Bhupinder Singh5,6 and John J Dillon7*
1Department of Cardiovascular Medicine, Mayo Clinic, USA
2Department of Cardiovascular Surgery, Mayo Clinic Ventures, USA
3Electrical and Computer Engineering, Ben-Gurion University of the Negev, Israel
4Division of Engineering, Mayo Clinic, USA
5Allakos Inc., 75 Shoreway Rd A, San Carlos, USA
6Division of Nephrology and Hypertension, University of California Irvine School of Medicine, USA
7Division of Nephrology and Hypertension, Mayo Clinic, USA
- *Corresponding Author:
- John J. Dillon
Division of Nephrology and Hypertension, Mayo Clinic, USA
Tel: 5072661046
E-mail: dillon.john@mayo.edu
Received date: August 17, 2018; Accepted date: August 29, 2018; Published date: September 03, 2018
Citation: Attia Z, Friedman PA, Ladewig DJ, Satam GC, Dugan JLF, et al. (2018) ECG-Based Potassium Measurement is Unaffected by Errors in Blood Potassium Measurement during Hemodialysis. J Clin Exp Nephrol .3:13. DOI: 10.21767/2472-5056.100064
Copyright: © 2018 Attia Z, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Background: Potassium abnormalities can cause lifethreatening arrhythmias. Measuring potassium requires access to blood. We have developed methods measuring potassium noninvasively using the processed, signalaveraged ECG. Four patients, in a larger study, were found to have unexpected discrepancies between measured blood potassium and ECG-derived estimated potassium values.
Methods: Of 240 patients enrolled at 17 sites in the PORTEND (REVEAL-HD) study, 200 wore a continuouslyrecording, single-lead, wireless ECG patch. Blood for chemistries was obtained once before, twice during and once after dialysis. Complete blood test and ECG data were available for 142 subjects. The general potassium pattern during dialysis was an exponential decay throughout the treatment. Four subjects, whose blood potassium values, but not ECG-based potassium values, deviated from this pattern, are the subjects of this analysis.
Findings: Among 4 patients, at least one blood potassium value declined to 2.6 mmol/l or less during dialysis, and then rebounded unexpectedly, while the ECG-based potassium values were consistent with the expected exponential delay. Three of these four patients were at a single site, suggesting site-specific likelihood of pattern deviation (p=0.04). In each case, BUN and phosphorous blood levels were markedly low, with albumin and calcium unaffected.
Conclusions: These results are compatible with blood drawing errors in which dialyzed blood was obtained from the venous return, rather than from the arterial tubing. A physiologic, ECG-based test that estimates potassium on the basis of the concentration of potassium in the blood surrounding the heart is free from local aberrations and might be a useful potassium monitoring tool in dialysis patients.
Keywords
Electrocardiography; Potassium; Kidney failure chronic; Dialysis
Introduction
Potassium homeostasis is essential to life, and hyper- or hypokalemia are associated with potentially life-threatening arrhythmias [1-4]. Patients with end-stage renal disease have potassium dysregulation, are prone to hyperkalemia, [5-7] with pre-dialysis hyperkalemia associated with increased sudden death, all-cause mortality and arrhythmia-related hospitalization [8]. This association raises the possibility that undetected and unmanaged hyperkalemia may contribute to the sudden death events, with perhaps the greatest risk occurring near the end of the three day weekend hiatus experienced by hemodialysis patients [6].
Currently, the only means of assessing serum potassium is through phlebotomy-based laboratory measurement of blood potassium, which requires travel to a medical facility, is painful, and requires laboratory equipment and trained personnel. In order to measure potassium levels at any time in a non-invasive way which can be controlled by the patients, ECG-based potassium assessments have been introduced. These tests may permit home-based remote potassium assessment, enhancing clinical care [9]. We recently obtained data from a multicentre cohort of patients undergoing haemodialysis while wearing a wireless ECG patch as part of a study to assess arrhythmia burden in dialysis patients [10]. While the ECG patch was not specifically designed or optimized for non-invasive potassium assessment, we applied the processing tools in an exploratory, developmental manner to compare blood potassium values derived from the processed single-lead ECG to the blood potassium test. In a subset of these patients, blood testing demonstrated a marked drop in potassium during dialysis followed by an atypical rebound as dialysis continued, in contrast to the typical exponential decay expected, whereas the physiological ECG-derived potassium test provided expected test results. We hypothesized that the unexpected blood test results stemmed from blood drawing errors during dialysis, leading to erroneous results, and that the ECG-derived potassium tests were unaffected, and more accurately reflected blood potassium levels.
Methods and Findings
Two hundred and forty patients were enrolled in the PORTEND (REVEAL-HD) study, designed to assess the interaction between potassium fluctuation and cardiac arrhythmias, and the burden of arrhythmias in dialysis patients. The main findings of that study have been reported elsewhere [10]. Briefly, study subjects wore a continuously-recording, single-lead, wireless ECG patch (BodyGuardian, Preventice, Minneapolis, MN) from study days one through five on dialysis days, and blood was obtained from the dialysis tubing immediately before dialysis, 30 min after the start of dialysis, 60 minutes after the start of dialysis, and immediately after dialysis. During dialysis, to prevent recirculation (dialyzed blood from the venous return being drawn into the arterial side of the dialysis circuit); blood was obtained after temporarily clamping the heparin line (if used for dialysis), halting the dialysis flow for 3 minutes and decreasing the blood flow rate to 100 cc/min for 15 seconds. Post-dialysis samples were obtained after decreasing the blood flow to 100 cc/min for 15 seconds. Blood samples were sent for potassium, calcium, phosphorous, creatinine, albumin, and BUN. There were no dietary restrictions or fasting requirements.
The single lead ECG was placed in a fixed position on the anterior chest for all five days, which was labelled with an indelible marker. The patch position was selected at the time of initial placement by sampling three standard precordial locations, and selecting the site with an optimal uniphasic twave. This ECG was used to estimate potassium, using techniques that we have described previously [9,11]. Using the ECG data from the first dialysis session and the acquired blood potassium test results, an individualized mathematical model describing the relationship between blood potassium and each patient’s ECG as acquired by the patch was automatically created in a Matlab framework. This model was used to determine blood potassium levels from the ECG data on subsequent dialysis days [9].
The sequential potassium blood tests acquired before, during and after hemodialysis were reviewed for each patient. The general pattern was an exponential decay, with most of the decline occurring early in the dialysis session, as seen previously by us and by others [9,11,12]. Four patients exhibited a pattern in which at least one blood potassium value fell to 2.6 or less during dialysis, and then rebounded by the end of dialysis. These results from these four patients were analyzed further by comparing them with the ECG-based potassium values and with the other blood constituent concentrations obtained from the same sample as the potassium measurements.
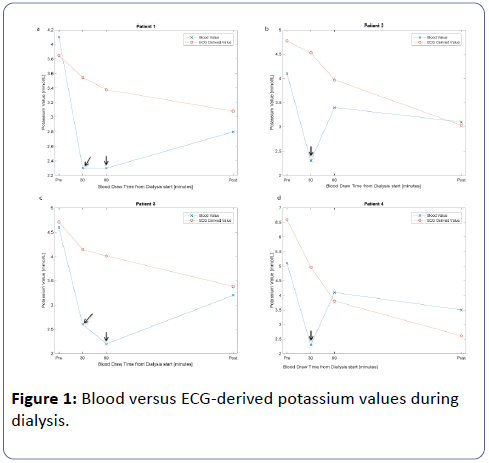
Of the two hundred and forty patients enrolled at 17 sites in the PORTEND study, ECG data were obtained in 200. Complete blood test results and ECG data were available in 142. Among these 142 patients, unexpectedly-low blood potassium values of 2.2-2.6 mmol/l were measured six times during dialysis among four patients (2.8% of total patients with complete data). Three of these four patients were at a single site, suggesting sitespecific likelihood (p=0.04 by Fisher’s Exact Test). Mean ± SD values for the four dialysis prescriptions were: duration 214 ± 31 min; blood flow rate 413 ± 48 ml/min; dialysate flow rate 694 ± 142 ml/min; dialysate potassium 2 ± 0 mmol/l and dialysate bicarbonate 33 ± 3 mmol/l. Three patients had arteriovenous fistulae and one had a central venous catheter. Subjects were instructed to continue their normal diets; there were no dietary restrictions or fasting requirements for the study. The sequential potassium blood test results and ECG-derived potassium estimates of these four patients are detailed in Figure 1. In each case, the ECG-derived potassium depicts a smooth, exponential decay, as expected during dialysis, while the blood values demonstrate a decline to very-low values, followed by a rebound. For the individual tests affected, the mean values of potassium (2.3 ± 0.1 mmol/l), BUN (3.1 ± 0.9 mg/dl) and phosphorous (0.7 ± 0.2 mg/dl) were markedly low, and creatinine was low for a dialysis patient (1.4 ± 0.9), while albumin (3.8 ± 0.4 g/dl) and calcium (8.7 ± 0.3 mg/dl) were unaffected.
This manuscript used existing, de-identified data and was deemed exempt from further review by the Mayo Clinic IRB.
Four patients demonstrated aberrantly-low blood potassium values during dialysis (arrows), a pattern not seen with the ECGderived potassium values.
Discussion
We found that blood potassium values, drawn during times at which the study protocol specified a pause in haemodialysis, were unexpectedly low, while ECG-derived potassium values were as expected, consistent with an exponential decay in serum potassium during haemodialysis. Moreover, phosphorus, BUN, and creatinine, substances removed by dialysis were also low, while albumin, which is not dialyzable, and calcium, which is dialyzable but has similar concentrations in blood and dialysate, was unaffected. These findings suggest that dialyzed blood, rather than non-dialyzed blood, was measured, suggesting that dialysis was not halted, as specified in the protocol and, moreover, that the affected samples were drawn from the venous return rather than from the arterial (pre-filter) side of the dialysis circuit. Additional possibilities for aberrantly low potassium values include recirculation and dilution of the blood sample. However, recirculation, which is usually less than 10%, [13] would have to be nearly complete to explain the potassium, BUN and phosphorus values in the affected samples. Dilution, for example if blood were obtained distal to an intravenous input or was diluted after the sample was obtained, would have affected the albumin and calcium values. Regardless of etiology, the ECG-derived potassium values, which measure the physiologic effect of potassium in the blood bathing cardiac tissue, indicate that the measured potassium values were not systemic.
There are other potential sources of error in blood potassium measurement. Haemolysis from excessive suction, drawing blood through a small needle or catheter or forcible ejection of the sample from a syringe may result in leakage of intracellular potassium [14-16]. Platelets and white blood cells may leak potassium into the serum before it is processed. If processing times are too short or too long, incomplete clotting, prolonged clot contact time or cellular metabolism may all modify test results [17]. Skin iodine (applied for sterile puncture) may increase potassium values if it contaminates the sample, [18] as can drawing blood through benzalkonium-heparin bonded catheters [14,15]. Even fist clenching, often erroneously performed prior to venipuncture, and may affect results. In one study comparing potassium drawn from a relaxed arm and from a fist clenched/pumped arm, fist pumping increased potassium values by up to 1.5 mmol/l, with a mean of 17% [19]. The mechanism is thought to be local release of muscle-cell potassium from the forearm muscles [20].
All potassium tests to date require blood. Recentlyintroduced, blood-free tests have estimated potassium from the signal processed ECG [9,11] and might be a reliable tool for monitoring patients during dialysis. In particular, with further validation, there is a possibility that, by identifying hypokalemia, hyperkalemia or excessive changes in potassium, these methods might be useful for preventing cardiac arrhythmias. ECG-based methods leverage the minute physiologic changes that occur with plasma potassium changes to estimate potassium blood levels. By eliminating the need for blood, they are amenable to remote potassium monitoring, which may permit virtually continuous and instantaneous monitoring of potassium, quality control of standard blood potassium measurements, and hence more-effective management of patients with renal disease, heart failure and diabetes. Since ECG-based measurements reflect physiologic change, they are not susceptible to mechanical or temperature, contamination, or the other potential errors associated with processing blood, but have other sources of potential error that may alter the ECG. These include lead-placement differences, changes in body position and changes in other electrolytes. With the development of robust signal processing techniques, these limitations can be overcome and in some cases may permit an accurate estimation of potassium when the blood tests are erroneous.
In conclusion, we present four cases in which errors in blood drawing resulted in falsely-low potassium measurements, while ECG-based measurements were as expected. A physiologic, ECGbased test that estimates potassium on the basis of the concentration of potassium in the blood surrounding the heart is free from local aberrations and might be a useful tool for monitoring potassium in dialysis patients.
Funding
The PORTEND (REVEAL-HD) study was funded by ZS-Pharma, Inc. Additional analysis of the patients presented in this study and the preparation of this manuscript was funded from internal Mayo Clinic funds.
Competing and Conflicting Interests
Mayo Clinic and authors ZA, PAF, DJL, GCS, JD, KEB, VKS and JJD have patents related to ECG-based potassium measurement. BS is a former employee of AstraZeneca, PLC. This research has been reviewed by the Mayo Clinic Conflict of Interest Review Board and is being conducted in compliance with Mayo Clinic Conflict of Interest policies.
References
- Einhorn LM, Zhan M, Hsu VD, Walker LD, Moen MF, et al. (2009) The frequency of hyperkalemia and its significance in chronic kidney disease. Arch Intern Med 169: 1156-1162.
- Juurlink DN, Mamdani MM, Lee DS, Kopp A, Austin PC, et al. (2004) Rates of hyperkalemia after publication of the Randomized Aldactone Evaluation Study. N Engl J Med 351: 543-551.
- Jain N, Kotla S, Little BB, Weideman RA, Brilakis ES, et al. (2012) Predictors of hyperkalemia and death in patients with cardiac and renal disease. Am J Cardiol 109: 1510-1513.
- Kovesdy CP, Regidor DL, Mehrotra R, Jing J, McAllister CJ, et al. (2007) Serum and dialysate potassium concentrations and survival in hemodialysis patients. Clin J Am Soc Nephrol 2: 999-1007.
- Bleyer AJ, Hartman J, Brannon PC, Reeves-Daniel A, Satko SG, et al. (2006) Characteristics of sudden death in hemodialysis patients. Kidney Int 69: 2268-2273.
- Perl J, Chan CT (2006) Timing of sudden death relative to the hemodialysis procedure. Nat Clin Pract Nephrol 2: 668-669.
- Wong MCG, Kalman JM, Pedagogos E, Toussaint N, Vohra JK, et al. (2015) Bradycardia and asystole is the predominant mechanism of sudden cardiac death in patients with chronic kidney disease. J Am Coll Cardiol 65: 1263-1265.
- Karaboyas A, Zee J, Brunelli SM, Usvyat LA, Weiner DE, et al. (2017) Dialysate Potassium, Serum Potassium, Mortality, and Arrhythmia Events in Hemodialysis: Results From the Dialysis Outcomes and Practice Patterns Study (DOPPS). Am J Kidney Dis 69: 266-277.
- Attia ZI, DeSimone CV, Dillon JJ, Sapir Y, Somers VK, et al. (2016) Novel Bloodless Potassium Determination Using a Signal-Processed Single-Lead ECG. J Am Heart Assoc 5: e002746.
- Singh B, Block G, Lerma EV, Tumlin J, Spinowitz B, et al. (2017) Hyperkalemia and Serum Potassium Variability in Patients on Hemodialysis. Am J Kidney Dis 65: A1-A3.
- Dillon JJ, DeSimone CV, Sapir Y, Somers VK, Dugan JL, et al. (2015) Noninvasive potassium determination using a mathematically processed ECG: proof of concept for a novel "blood-less, blood test". J Electrocardiol 48: 12-18.
- Agar BU, Culleton BF, Fluck R, Leypoldt JK (2015) Potassium kinetics during hemodialysis. Hemodial Int 19: 23-32.
- Tan J, Mohan S, Herbert L, Anderson H, Cheng JT (2012) Identifying hemodialysis catheter recirculation using effective ionic dialysance. ASAIO J 58: 522-525.
- Baer D, Ernst D, Willeford S, Gambino R (2006) Investigating elevated potassium values. Medical Laboratory Observer 24-31.
- Stankovic AK, Smith S (2004) Elevated serum potassium values: the role of preanalytic variables. Am J Clin Pathol 121: S105-S112.
- Bailey IR, Thurlow VR (2008) Is suboptimal phlebotomy technique impacting on potassium results for primary care? Ann Clin Biochem 45: 266-269.
- Nijsten MW, de Smet BJ, Dofferhoff AS (1991) Pseudohyperkalemia and platelet counts. N Engl J Med 325: 1107.
- Van Steirteghem AC, Young DS (1977) Povidone-iodine ("Betadine") disinfectant as a source of error. Clin Chem 23: 1512.
- Don BR, Sebastian A, Cheitlin M, Christiansen M, Schambelan M (1990) Pseudohyperkalemia caused by fist clenching during phlebotomy. N Engl J Med 322: 1290-1292.
- Asirvatham JR, Moses V, Bjornson L (2013) Errors in potassium measurement: a laboratory perspective for the clinician. N Am J Med Sci 5: 255-259.
Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences