Different Effects of EP2 and EP4 Receptors in TGF-ÃÆà ½Ãâò1 Induced Mesangial Cells Injury
Xiaolan Chen, Jing Wang, Hu Xu, Tianyi Pan, Yuying Xu, Xu Chen, Yaping Fan
DOI10.21767/2472-5056.100018
Tianyi Pan1,Wang Jing1,2*, Hu Xu1,3, Xiaolan Chen1*,Yuying Xu1, Xu Chen1 and YapingFan1
1Department of Nephrology, Affiliated Hospital of Nantong University, Nantong, Jiangsu, PR China
2Shanghai Jiaotong University, School of Medicine, Shanghai, PR China
3Peking University, School of Medicine, Beijing, PR China
- *Corresponding Author:
- Xiaolan Chen
Department of Nephrology, Affiliated Hospital of Nantong University
Nantong, Jiangsu, PR China
E-mail: chenxl8448@sina.com
Jing Wang
Shanghai Jiaotong University, School of Medicine
Shanghai, China
E-mail: jingsophie@vip.sina.com
Received date: July 05, 2016; Accepted date: August 10, 2016; Published date: August 12, 2016
Citation: Chen X, Wang J, Xu H, Pan T, Xu Y, et al. (2016) Different Effects of EP2 and EP4 Receptors in TGF-β1 Induced Mesangial Cells Injury. J Clin Exp Nephrol 1:18. DOI: 10.21767/2472-5056.100018
Copyright: © 2016 Chen X, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Background: The prostaglandin E2 (PGE2) and its receptor subtypes 2 and 4 (EP2 and EP4) in mesangial cells (MCs) proliferation and glomerulosclerosis hasn’t been elaborated in detailed.
Methods: We cultured EP2-/-, EP4flox/flox, and WT mice mesangial cells to detect the relevant indicators and downstream signalling. Additionally, 5/6 nephrectomy were introduced to confirm the in vitro outcomes.
Results: We found that the expression of fibronectin (FN), COX-2 and the phosphorylation level of Smad3 in group EP2-/- obviously increased, with the falling of cyclic adenosine monophosphate (cAMP) level and no protein kinase (PKA) activity. While the relevant genes decreased in EP4 knock out (EP4flox/flox + AD-CRE) group, which represent no significant change in the level of cAMP, although with high level of PKA activity and extracellular signal transduction kinase (ERK) phosphorylation. In vivo study, 24-hour proteinuria, serum creatinine and urea nitrogen in EP4+/- group obviously declined, and the degree of glomerular sclerosis and the density of FN and COX-2 in glomerulus reduced significantly. EP2-/- show a totally opposite event to EP4+/- .
Conclusion: EP2 receptor mainly depends on cAMP/PKA to inhibit effect of PGE2 induced by TGF-β1/Smad3 pathway and further alleviate the damage of MCs. While EP4 may coordinate the effect of MCs injury through ERK phosphorylation. Our finding suggests the pleiotropic functions of PGE2, and thus explores a potential therapy on the level of gene mediation.
Keywords
Renal fibrosis; Prostaglandin; E2 receptor subtypes 2 and 4; Gene knockout; Mouse mesangial cells; Extracellular signal-regulated kinase (ERK); 5/6 nephrectomy
Introduction
Renal fibrosis, the main cause to CKD (chronic kidney disease), play a pivotal role in the progression of ERSD (end renal stage disease). The introduction of cytokines, such as TGF-β1, promotes PGE2 secretion as well as the occurrence of tissue fibrosis [6,7]. Cyclooxygenase 2 (COX-2) derived PGE2 can act on [8] EP1, EP2, EP3 and EP4, four functional receptors of PGE2, all of which are G protein coupled septic-transmembrane ones [9], to mediate the pathophysiologic procedures. EP2 and EP4 can connect with stimulation G (Gs) protein to increase the level of cAMP in different degrees. While EP4 can also couple with inhibitory G (Gi) protein to activate MAPKs [10]. According to the location research, EP4 is positioned in both glomerulus and peritubular capillary. Although the quantities of EP2 in glomerulus are relatively small, it was reported that after mice were subjected to subtotal nephrectomy, the manifestation of EP2 receptor obviously increased [11]. All the previous studies indicate that the two receptors play a vital role in renal disease.
In our research, we took mesangial cells as the target to determine the role of EP2 and EP4 receptor in glomerulus damage, as extracellular matrix (ECM) initially forms in mesangium and ECM associated indicators abundantly expressed [12]. Through intervening the PGE2 pathway, we might detect a novel potential treatment to glomerular sclerosis enhanced by MCs proliferation to reverse the CKD progression.
Results
Endogenous PGE2 secretion stimulated by TGF-β1
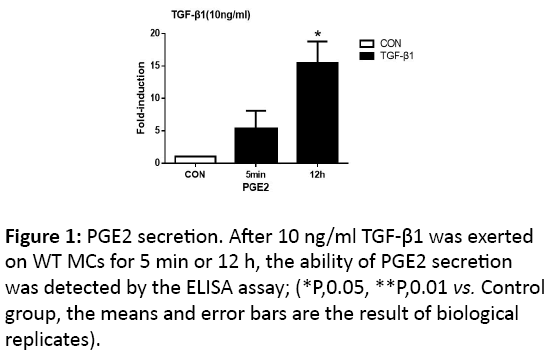
With the presence of 10 ng/ml TGF-β1 on MCs for 5 min and 12 h respectively, secretion of PGE2 could facilitate, and the change in latter time point was of statistical significance (p<0.05) (Figure 1).
The knockout of EP2 and EP4 receptors
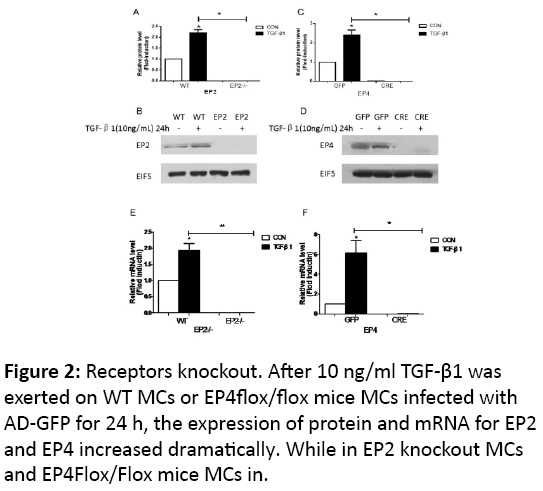
We established related gene knockout mice, and examined the receptors’ expression with 10 ng/ml TGF-β1 for 24 h, the expression of protein and mRNA of the two receptors increased mildly (p<0.05). While in knock out groups, no matter with the stimulation by TGF-β1, there is no evidence to show the expression of the protein, as well as the mRNA (Figure 2).
The expression of related fibrosis indicators
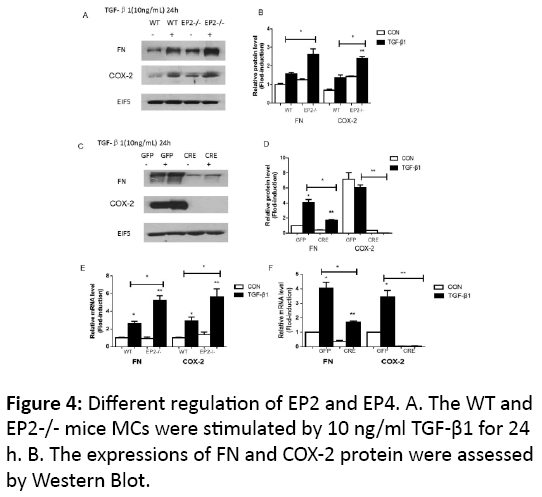
After the stimulation of 10 ng/ml TGF-β1 in WT MCs for 24 h, mRNA and protein level of FN and COX-2 increased obviously (p<0.05)(Figure 3), and EP2 receptor knocking out enhanced the expression of the related mRNA and protein (p<0.05) (Figures 4A, 4B and 4C). The transfection of AD-CRE to EP4flox/flox MCs weaken the mRNA and protein level of both genes, compared with EP4flox/flox + AD-GFP group (p<0.05, p<0.01) (Figures 4D, 4E and 4F).
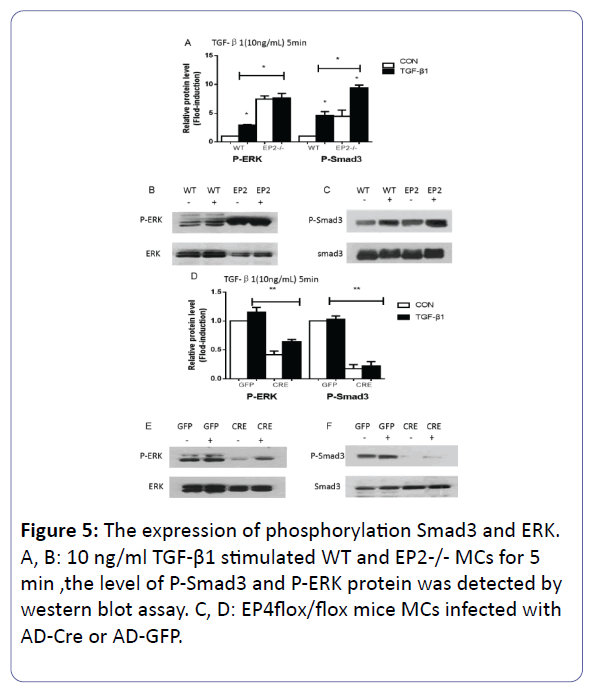
The phophrylation of Smad3 and ERK
Each group were stimulated by TGF-β1 with 10 ng/ml for 5 min to strengthen the average level of the phosphorylation of the signaling (p<0.05), however, the difference between the group EP4flox/flox + AD-GFP and the group EP4flox/flox + ADGFP +TGF-β1 appeared with no statistically significance on the expression of both genes. Compared with WT + TGF-β1, the amount of the protein of P-Smad3 and P-ERK increased about 2 times in EP2-/- + TGF-β1 MCs (p<0.05) (Figures 5A, 5B and 5C). Adversely, the EP4 knock out were involved in the inhibition of the phosphorylation of Smad3 and ERK (p<0.05) (Figures 5D, 5E and 5F).
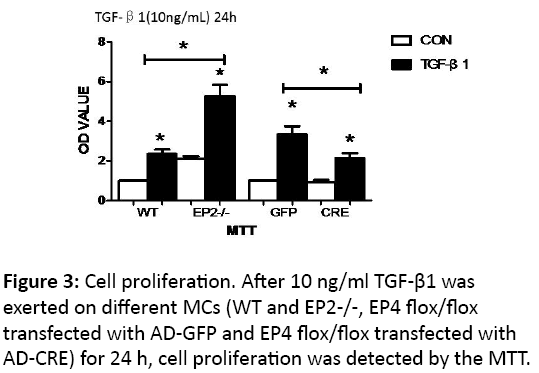
The proliferation of mesangial cells
The proliferation of mesagial cells was detected by MTT assay and measured with OD values.
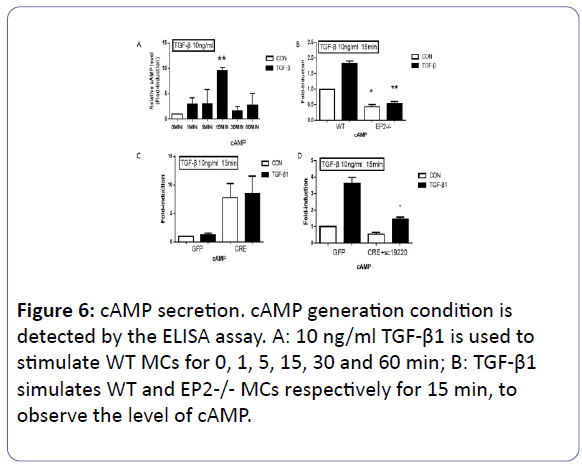
The camp level
After 10 ng/ml TGF-β1 was used for stimulating WT MCs for 0, 1, 5, 15, 30 and 60 min respectively, the variation of cAMP level was measured by the ELISA assay. Compared to the control group, with the appearence of TGF-β1 for 15 min, the value of cAMP in WT MCs reached the peak (p<0.01) (Figure 6A). Subsequently, we carried the expriement on the different groups for 15 min. The EP2 receptor has the priority in regulation of the level of cAMP, compared with the WT group (p<0.05). The ADCRE added to the EP4flox/flox MCs changed the level of cAMP with no statistically significance (P>0.05) (Figures 6B & 6C). To futher confirm the essencial role of the EP2 receptor for cAMP level, 1mm EP2 antagonist (SC-19220) was added in EP4flox/flox + AD-CRE group to completely exclude the influence of EP2, and the cAMP synthesis capability in this group reduced obviously(p<0.05) (Figure 6D).
PKA activity detection
The activity of PKA in EP2-/- MCs was detected by Peptag nonradioactive kit indicated that no matter being stimulated by 10 ng/ml TGF-β1(stimulation time: 15 min), there is no activity, compared to the high level of that in WT group (Figure 7A). The (Figure 7B) told us that the deficiency of the EP4 inhibited the activity of PKA, but the addition of the 10 ng/ml TGF-β1 recreated the activity, compared to the EP4flox/flox +AD-GFP group with continuously PKA activity.
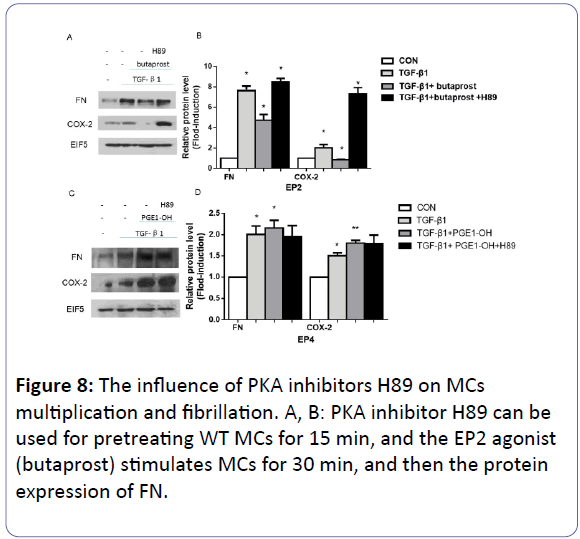
The inhibition effects of H89
1 mM H89 was introduced for pre-treating WT MCs for 15 min to block the PKA activity, followed by the addition of 10 mM EP2 agonist (butaprost) or 0.1 mM EP4 agonist (PGE1-OH) for 30 min respectively. While the DMSO was given to the control group. Then the serum-free culture medium was changed, 10 ng/ml TGF-β1 appeared to treat each dish of MCs for 24 h. In our findings, with TGF-β1, the protein expression of FN and COX-2 improved obviously compared with the control group (p<0.05). Secondly, although there was reduction of the two genes at the presence of butaprost (p<0.05), the addition of H89 reverse the protein level (p<0.05) (Figures 8A and 8B). Moving to the graph below, we can see that PGEI to active the EP4 enhanced the expression of FN and COX-2 protein, the introduction of H89 made no significant change yet (p>0.05) (Figures 8C and 8D).
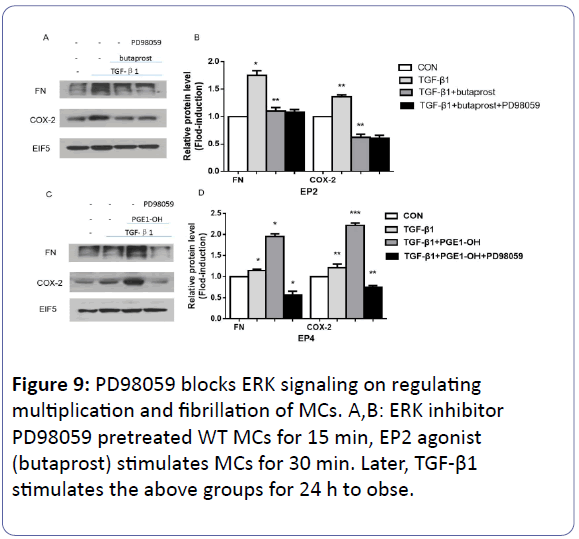
The inhibition effects of PD98059
The effect of EP4 on ERK signaling was detected with the 1 mM PD98059 (ERK inhibitor) pre-treating WT MCs for 15 min, followed by the addition of 10 mM EP2 agonist (butaprost) or 0.1 mM EP4 agonist (PGE1-OH) for 30 min respectively. Subsequently, 10 ng/ml TGF-β1 was introduced (Figures 9A and 9B). The results indicates that after TGF-β1 was exerted on the above groups for 24 h, compared with the control group, the expression of FN and COX-2 reduced obviously after butaprost activates EP2 receptor (p<0.05). However, PD98059 did not work in this group (p<0.05) (Figures 9C and 9D). On the contrary, PD98059 can block that kind of increasing by PGEI in EP4 (p<0.05).
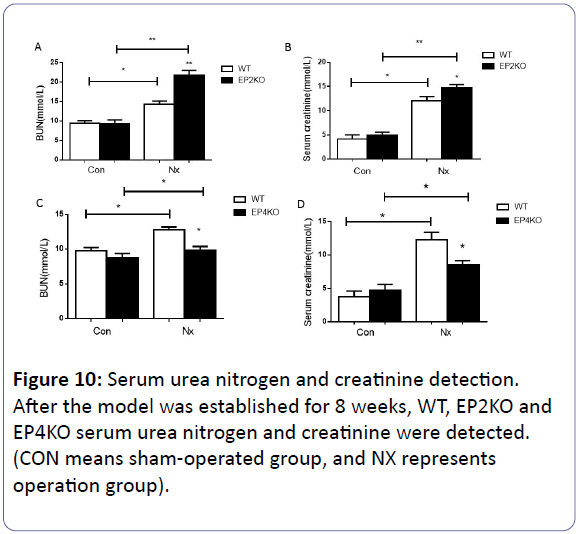
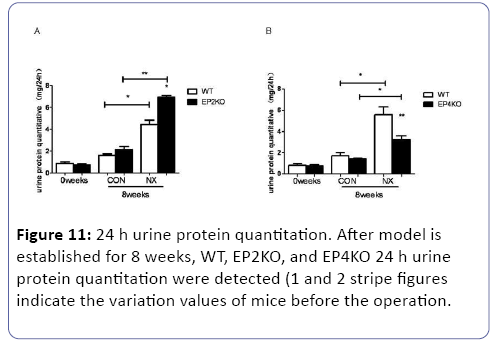
The quantitation of 24 h urine protein, serum creatinine and urea nitrogen
Each group of mice were executed after subtotal nephrectomy for 8 weeks. We examined the 24 h urine protein quantitation, serum urea nitrogen (BUN) and serum creatinine (CR) in vivo study. Once being subjected to the subtotal nephrectomy for 8 weeks, the 24 h urine protein, BUN and CR increased obviously (p<0.05) compared with pre-operation or sham operation mice. Compared with WT NX group, EP2-/- NX mice 24 h urine protein, BUN and CR saw a dramatic rise. (p<0.05) (Figures 10A, 10B, and 11). On the contrary, the EP4 knock out caused a reduction of the three related indexes (p<0.05) (Figures 10C, 10D, and 11).
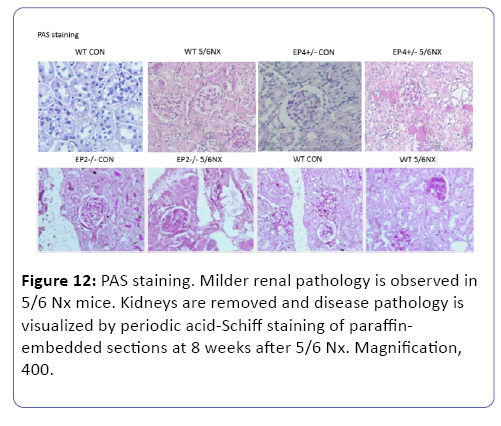
Pathological variation after 5/6 nephrectomy
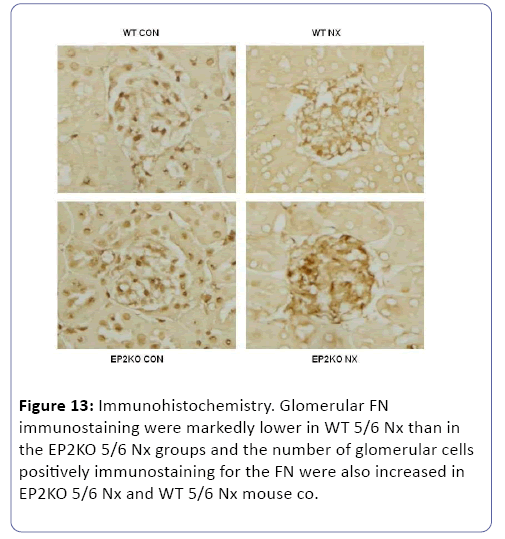
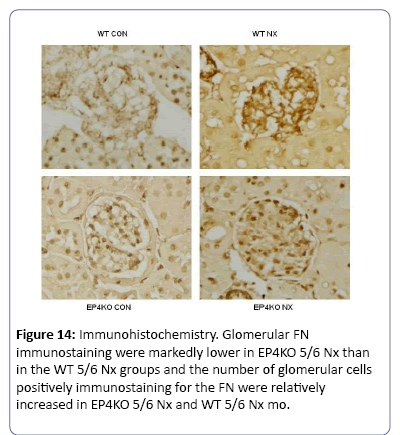
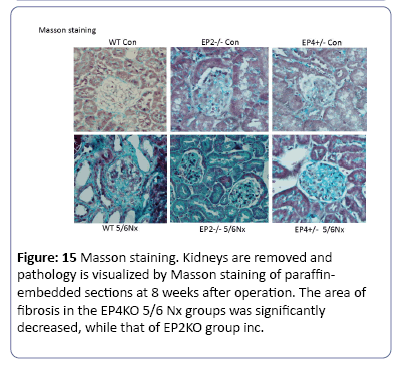
The mice subjected to subtotal nephorectomy showed greater pathological changes than those in the sham operation. Compared to WT group, the mice in EP2KO group after operation for 8 weeks suffered from heavier adhesion between Bowman’s capsule and gromerular, and a diffusive proliferation of glomerular mesangial cells and mesangial matrix among capillary loops. While the mesangial proliferation was not obvious in EP4KO group (Figure 12). Immunohistochemistry was used to observe the density of FN deposited in gromerulus in each group, the mice suffered from operation presented higher levels of FN deposition than those in non-operation group (P<0.01). Compared with WT group, FN density in EP2KO group increased less than 5 folds (P<0.05). (Figures 13 and 14), (Table 1). The Masson staining showed that there was less collagen deposition (blue area) around the renal glomerulus in the EP4+/- groups, while the area of fibrosis in EP2-/- 5/6 Nx group increased dramatically, both compared to WT group (Figure 15).
| Primers For Real-Time PCR Analysis | |||
|---|---|---|---|
| Gene Name | Chain | Sequence(5’-3’) | Product (bp) |
| GADPH | FP | AGAAGGCTGGGGCTCATT TG | 238 |
| RP | AGGGGCCATCCACAGTCT TC | ||
| FN | FP | AATGGAAAAGGGGAATGG AC | 244 |
| RP | CTCGGTTGTCCTTCTTGCT C | ||
| COX-2 | FP | CAAAGCAGCTGCAAATAC CA | 220 |
| RP | GGCCAAATGTGTCTTCCA GT | ||
Table 1: The primer sequences are in PCR products were validated by electrophoresis on 2% agarose gel.
Methods and Materials
Experiment animals
WT (wild-type), EP2 homozygotes(EP2-/-), EP4 heterozygotes (EP4+/−) and EP4flox/flox (with a conditional knock-out EP4 gene sequence between LoxP sites as the sites flanking exon 2 of the EP4 gene) male mice aged 8-12 weeks were selected in the experiment. The latter was subject to transfection with CRE adenovirus to knock out EP4 receptors [13]. The mice were euthanized with an overdose of diethyl ether.
Adenoviral constructs and infection of cultured mice mesangial cells
The AD-Cre was generated by the Shanghai GenePharma Co. Ltd. Linearized recombinant adenoviral plasmid was transfected into AD-293 cells to obtain a primary viral stock, which was amplified and purified. For optimization of infection conditions, differentiated mouse MCs were infected with AD-Cre at a 10 multiplicity of infection (MOI) for 72 h. Expression of EP4 in infected WT MCs was examined by Western blot.
Culuture of primary mesangial cells (MCs)
The specific method of MCs culture was according to the protocol of our previous experiment [14].
ELISA asssy
The supernatant of the samples in each groups were collected, in which The quantity of cAMP was detected using an Alpha screen cAMP Assay Kit (Perkin Elmer, Massachusetts, USA).Similarly the PGE2 level were also measured with an Alpha screen PGE2 Assay Kit, while the test just happened in WT cells, and such cells stimulated by TGF-β1 for 5 min or 12 h. Both these tests were according to the manufacturer’s instructions, and read by an EnVision Multilabel Plate Reader.
PepTag nonradioactive PKA activity detection
PepTag non-radioactive protein kinase kit is used for detecting intracellular PKA activity in different groups.
5/6 nephrectomy
40 mice weight of 25-35 g, with the background of c57bl/6 are adopted in the experiment, are limited in eating and drinking before the night of surgery. The 5/6 nephorectomy were held according to our previous study, and some details and process were recorded the relevant protocol [15]. Groups of the experiment are divided into WT CON group, WT NX group; EP2-/- (EP2KO) CON group , EP2-/- (EP2KO) NX group; EP4+/- (EP4KO) CON group, EP4+/- (EP4KO) NX group. The mice were executed with overdose of diethyl ether after 8 weeks, the serum and residual kidney is collected for the measurement of serum creatinine, urea nitrogen and urine protein. Meanwhile, pathological examination is conducted, and left tissues and serum are reserved at -80.
Renal histological assay
The specific method was according to the protocol of our previous experiment [16].
Immunohistochemistry
The expression of fibronectin (FN) was detected using immunohistochemical staining methods. Polyclonal rabbit antibodies against FN was used (Cayman, USA).
Statistical analysis
All values are expressed as mean ± SEM. Data were analyzed by Student t test (paired groups) or 2-way ANOVA, followed by Bonferroni’s post-test correction (multi group comparisons). P<0.05 was considered statistically significant.
Discussion
Many researches testify that PGE2 has important effects on regulation of renal diseases, while the specific role on mesangial cells and glomerular damage hasn’t been clearly discussed [15,16]. PGE2 show the pleiotropic nature through activating four subtypes [17]. In present study, we cultured EP2-/- mesangial cells, as EP4-/- mice always suffer patent ductus arteriosus postnatally, special EP4 knockout ones were established, which has been used in our previuos studies [14], to explore the role of the two receptors in mesangial cells injury by TGF-β1. Our results suggested that EP4 knock out might show synergetic injury effect different from the protective role of EP2 in mesangial cells damage.
PGE2 working on the receptors coupled Gs protein always activates adenylate activating enzyme (AC), followed by the increase of cAMP levels and activation of PKA [18]. Studies have found that, in pulmonary artery smooth muscle cells, bradykinin can promote the formation of cAMP, and this function is associated with EP2 and EP4 receptor activation [19]. Therefore, cAMP/PKA may perform as a linkage between the proliferation
of MCs and the two receptors. Our further experiments demonstrated that EP2 knockout obviously promote the formation of cAMP/PKA in mesangial cells, which alleviates its proliferation induced by TGF-β1. A study reported that, although there is an almost equal amount of EP4 and EP2 in the same type of cells, the activation of EP4 receptor had a limited contribution to the expression of cAMP [20], consistent with our current results. EP4 may regulate the accumulation of extracellular matrix on mesangium through other pathways.
Extracellular signal regulated kinase (ERK) carries the information from the surface to the nucleus, and is able to be activiated by different stimuli [21]. In addition, it can have a crosstalk with smads pathway in renal fibrosis: phosphorylation of Smad1, Smad2 and Smad3 by Ras-activated ERK1/2 in the linker region leads to the inhibition of their nuclear translocation [22]. Besides, Studies have found that EP4, coupling with inhibitory G (Gi) protein, phosphorylated ERK signaling and regulated gene transduction [23]. Quan He et al found that inhibition of PGE2 production reduces hypertrophy after myocardial infarction in mice and PGE2 through its EP4 receptor causes hypertrophy of neonatal ventricular myocytes via ERK1/2 phosphorylation [24]. All these reports combine with our results; prove that the phosphorylation of ERK may be involved in the co-injury effects of PGE2/EP4 in mesangium related glomerulosclerosis induced by TGF-β1.
We also introduced butaprost (EP2 agonist) and PGE1-OH (EP4 agonist) respectively to comfirm our findings. Both two compounds has been used in many previous researches to testify their efficiency [25]. In present studies, EP2 agonist successfully down-regulates the expression of FN and COX-2, while EP4 agonist up-regulate them. The former effect was affected by H89, a specific PKA inhibitor, which can enhance angiotensin II-induced superoxide generation, and further upregulate mRNA levels of intercellular adhesion molecule-1 and plasminogen activator inhibitor-1 in mesangial cells to promote the cells injury [26]. PD98059 (ERK inhibitor) block EP4 effectively [27]. We believe that TGF-β1 activates EP4 receptor to induce ERK phosphorylation and thereby increase mesangial cells proliferation.
Although EP2 and EP4 receptor inhibit progression of fibrosis cooperatively or independently in the liver, lungs and other tissues [28]. Nevertheless, many current studies show the role of EP4 receptor is still controversial [29,30]. In our in vivo studies, we built three genotypes of mice (WT, EP2-/-, EP4+/-) to underwent subtotal nephrectomy respectively. EP4 comfirmed its maladaptive role. In podocytes, the mice with specific over expression of EP4 receptor accepted the subtotal nephrectomize. After 2 weeks, the amount of the survival decreased a lot along with the high level of serum creatinine and urinary protein [31]. Therefore, the role of EP4 in kidney would depend on the species of cells and tissues, and partly are determined by the location of the receptors, also is associated with the current stages of disease development [32,33].
Briefly, EP2 receptor stimulates cAMP/PKA pathway, and inhibits proliferation of mesangial cells triggered by TGF-β1/ Smad3 and synthesis of extracellular matrix and COX-2 [34]. However, the activation of EP4 enhances the phosphorylation of downstream ERK signaling, thus the damage of mesangial cells induced by the TGF-β1/Smad3 can be amplified. We understand that pathological change of whole kidney will better reveal the significance of the two receptors. But in present study, we mainly focused on gromerular injury, and our results should be sufficient to draw a conclusion that EP2 and EP4 actually show different effect on the proliferation of mesangial cells and glomerular damage. To sum up, as the deficient of effective therapy to reverse the progression of CKD, the intervention on the gene level may be a novel treatment.
Compliance with Ethical Standards
Ethics statement
The experimental animals were provided by the Animal Experimentation Committee of Peking University Health Science Center according to SPF environment, with a gene background of C57/BL6. Animal protocols for these studies were supported by the Peking University Animal Care and Use Committee.
Conflict of interest
The authors have declared that no conflict of interest exists.
References
- Liu Y (2006) Renal fibrosis:New insights into the pathogenesis and therapeutics. Kidney Int 69: 213-217.
- Li W, Cui M, Wei Y (2012) Inhibition of the Expression of TGF-β1 and CTGF in Human Mesangial Cells by Exendin-4, a Glucagon-like Peptide-1 Receptor Agonist. Cell Physiol Biochem 30: 749-757.
- Meng XM, Huang XR, Chung ACK (2010) Smad2 protects against TGF-β/Smad3-mediated renal fibrosis. J Am Soc Nephrol 21: 1477-1487.
- Sato M, Muragaki Y, Saika S (2003) Targeted disruption of TGF-β1/Smad3 signaling protects against renal tubulointerstitial fibrosis induced by unilateral ureteral obstruction. J Clin Invest 112: 1486-1494.
- Bondi CD, Manickam N, Lee DY (2010) NADPH oxidase mediates TGF-β1–induced activation of kidney myofibroblasts. J Am Soc Nephrol 21: 93-102.
- López-Hernández FJ, López-Novoa JM (2012) Role of TGF-β in chronic kidney disease: an integration of tubular, glomerular and vascular effects. Cell Tissue Res 347: 141-154.
- Ivanova V, Garbuzenko OB, Reuhl KR (2013) Inhalation treatment of pulmonary fibrosis by liposomal prostaglandin E2. Eur J Pharm Biopharm 84: 335-344.
- Bataller R, Ginès P, Nicolás JM (2000) Angiotensin II induces contraction and proliferation of human hepatic stellate cells. Gastroenterology 118: 1149-1156.
- Suzawa T, Miyaura C, Inada M (2000) The role of prostaglandin E receptor subtypes (EP1, EP2, EP3, and EP4) in bone resorption: an analysis using specific agonists for the respective EPs. Endocrinology 141: 1554-1559.
- Morath R, Klein T, Seyberth HW (1999) Immunolocalization of the four prostaglandin E2 receptor proteins EP1, EP2, EP3, and EP4 in human kidney. J Am Soc Neph 10: 1851-1860.
- Schnaper HW, Hayashida T, Hubchak SC (2013) TGF-β signal transduction and mesangial cell fibrogenesis. Am J Physiol Renal Physiol 284: F243-F252.
- Breyer MD, Breyer RMG (2001) protein-coupled prostanoid receptors and the kidney[J]. Annu Rev Physiol 63: 579-605.
- Schneider A, Guan YF, Zhang Y (2004) Generation of a conditional allele of the mouse prostaglandin EP4 receptor. Genesis 40: 7-14.
- Yang G, Xu Y, Fan Y (2014) A Maladaptive Role for EP4 Receptors in Mouse Mesangial Cells. PloS One 9: e104091.
- Nasrallah R, Hassouneh R, Hebert RL (2014) Chronic kidney disease: targeting prostaglandin E2 receptors. Am J Physiol Renal Physiol 307: F243-F250.
- Ito S, Carretero OA, Abe K (1989) Effect of prostanoids on renin release from rabbit afferent arterioles with and without macula densa. Kidney Int 35: 1138-1144.
- Kawahara K, Hohjoh H, Inazumi T (2015) Prostaglandin E 2-induced inflammation: Relevance of prostaglandin E receptors. Biochim Biophys Acta 1851:414-421.
- Insel PA, Murray F, Yokoyama U (2012) cAMP and Epac in the regulation of tissue fibrosis. Br J P harmacol 166: 447-456.
- Bradbury DA, Newton R, Zhu YM (2003) Cyclooxygenase-2 induction by bradykinin in human pulmonary artery smooth muscle cells is mediated by the cyclic AMP response element through a novel autocrine loop involving endogenous prostaglandin E2, E-prostanoid 2 (EP2), and EP4 receptors. J Biol Chem 278: 49954-49964.
- Fujino H, West KA, Regan JW (2002) Phosphorylation of glycogen synthase kinase-3 and stimulation of T-cell factor signaling following activation of EP2 and EP4 prostanoid receptors by prostaglandin E2. J Biol Chem 277: 2614-2619.
- Tian W, Zhang Z, Cohen DM (2000) MAPK signaling and the kidney [J]. Am J Physiol Renal Physiol 279: F593-F604.
- Kretzschmar M, Doody J, Timokhina I, Massague J (1999) A mechanism of repression of TGFbeta/Smad signaling by oncogenic Ras. Genes Dev 13: 804-816.
- Fujino H, Xu W, Regan JW (2003) Prostaglandin E2 induced functional expression of early growth response factor-1 by EP4, but not EP2, prostanoid receptors via the phosphatidylinositol 3-kinase and extracellular signal-regulated kinases. J Biol Chem 278: 12151-12156.
- Quan He, Harding P, Margot C (2010) PKA, Rap1, ERK1/2, and p90RSK mediate PGE2 and EP4 signaling in neonatal ventricular myocytes.Am J Physiol Heart Circ Physiol 298: 136-143.
- Ishibashi R, Tanaka I, Kotani M (1999) Roles of prostaglandin E receptors in mesangial cells under high-glucose conditions. Kidney Int 56: 589-600.
- Ishibashi Y, Matsui T, Ojima A (2012) Glucagon-like peptide-1 inhibits angiotensin II-induced mesangial cell damage via protein kinase A. Microvasc Res 84: 395-8.
- Li XC, Carretero OA, Shao Y (2006) Glucagon receptor-mediated extracellular signal-regulated kinase 1/2 phosphorylation in rat mesangial cells: role of protein kinase A and phospholipase C. Hypertension 47: 580-5.
- Bozyk PD, Moore BB (2011) Prostaglandin E2 and the pathogenesis of pulmonary fibrosis. Am J Respir Cell Mol Biol 45: 445-452.
- Vukicevic S, Simic P, Borovecki F (2006) Role of EP2 and EP4 receptor-selective agonists of prostaglandin E2 in acute and chronic kidney failure. Kidney Int 70: 1099-1106.
- Mohamed R, Jayakumar C, Ramesh G (2013) Chronic administration of EP4-selective agonist exacerbates albuminuria and fibrosis of the kidney in streptozotocin-induced diabetic mice through IL-6. Lab Invest 93: 933-945.
- Stitt-Cavanagh EM, Faour WH, Takami K (2010) A maladaptive role for EP4 receptors in podocytes. J Am Soc Nephrol 21:1678-1690.
- Nakagawa N, Yuhki K, Kawabe J (2012) The intrinsic prostaglandin E2–EP4 system of the renal tubular epithelium limits the development of tubulointerstitial fibrosis in mice. Kidney Int 82: 158-171.
- Boor P (2012) EP4: a new piece in the fibrotic puzzle [J]. Kidney Int 82: 132-135.
- Xu Y, Wang J, Pan T (2016) Role of the ER stress in prostaglandin E2/E-prostanoid 2 receptor involved TGF-β1 induced mice mesangial cell injury. Mol Cell biochem 411: 43-55.
Open Access Journals
- Aquaculture & Veterinary Science
- Chemistry & Chemical Sciences
- Clinical Sciences
- Engineering
- General Science
- Genetics & Molecular Biology
- Health Care & Nursing
- Immunology & Microbiology
- Materials Science
- Mathematics & Physics
- Medical Sciences
- Neurology & Psychiatry
- Oncology & Cancer Science
- Pharmaceutical Sciences